Hepatitis E virus (HEV) remains a major cause of acute viral hepatitis worldwide, with an estimated 20 million infections annually. Although infection is often self-limiting, severe disease occurs in immunocompromised individuals and pregnant women. Despite its global burden, no specific antiviral therapy is currently available.
A major obstacle in HEV research has been the limited understanding of how the virus hijacks host machinery to support its replication. The viral replicase, encoded by ORF1, forms a large and dynamic replication complex that depends heavily on host factors. However, the lack of robust experimental systems and suitable small animal models has long hindered systematic identification of essential host components.
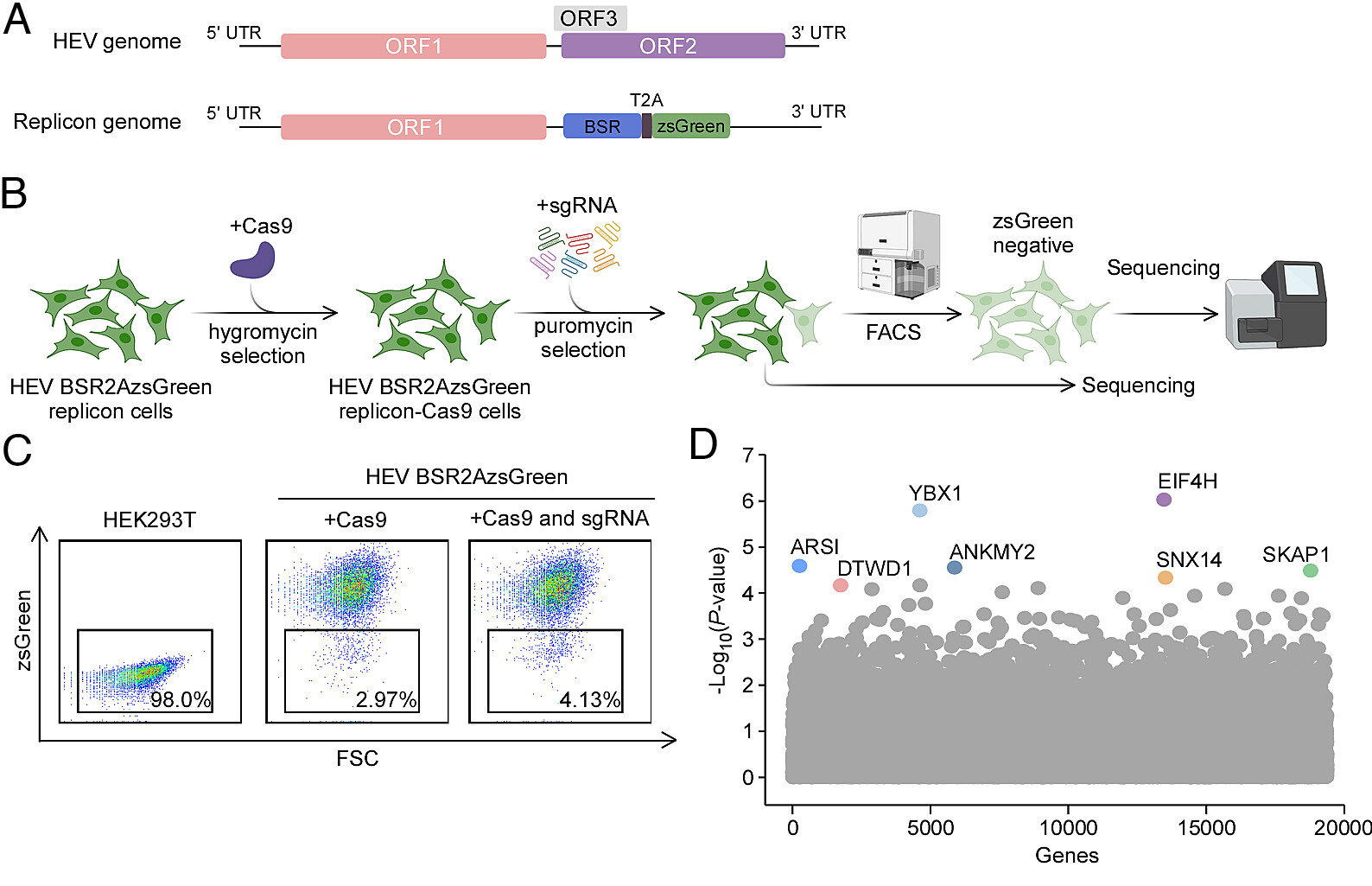
In a recent study published in PNAS, Dr. Qiang Ding (Tsinghua University) and colleagues, in collaboration with Dr. Lin Wang (Peking University) and international partners, report two critical host factors: EIF4H and YBX1, that are indispensable for HEV replication and pathogenesis.
To overcome longstanding technical barriers, the team first established a stable HEV replicon system and performed an unbiased genome-wide CRISPR screen to identify host genes required for viral replication. The screen revealed EIF4H and YBX1 as key host dependency factors. Mechanistic analyses demonstrated distinct roles for these proteins: EIF4H facilitates efficient viral RNA synthesis by supporting the assembly and function of the viral replication complex. YBX1 regulates the processing and maturation of the ORF1 polyprotein, thereby influencing proper assembly of the viral replication machinery. These findings provide new insight into how HEV coordinates host translational and RNA-binding factors to sustain replication.
To establish physiological relevance, the team generated EIF4H- and YBX1-deficient rat models. Upon HEV infection, knockout animals displayed markedly reduced viral replication and fecal shedding, accompanied by significantly attenuated liver pathology. These in vivo genetic data provide compelling evidence that EIF4H and YBX1 are not only required for replication in cell culture, but also play critical roles in viral infection and disease progression.
This study advances understanding of HEV replication biology and pathogenesis. Moreover, EIF4H and YBX1 represent potential new targets for host-directed antiviral strategies, an especially attractive approach given the limited therapeutic options currently available.
This work exemplifies the power of combining genome-wide functional screening and genetically engineered animal systems to dissect RNA virus-host interactions. It also provides a valuable framework for future efforts aimed at targeting host dependency factors in emerging viral infections.
Read the full article: Proc Natl Acad Sci U S A. 2026 Mar 10;123(10):e2529289123. DOI: 10.1073/pnas.2529289123