Hepatitis E virus (HEV) is a major cause of acute hepatitis globally, yet the mechanisms underlying its replication and pathogenesis remain poorly understood. While mitochondrial dynamics—the balanced processes of fission and fusion—are increasingly recognized as central regulators of cellular defense and antiviral responses, their role in HEV infection has not been defined.
A recent study led by Dr. Yijin Wang and PhD candidate Liu Xiaoman of the Southern University of Science and Technology, China, demonstrated that HEV actively induces mitochondrial fusion, a process essential for optimal viral replication.
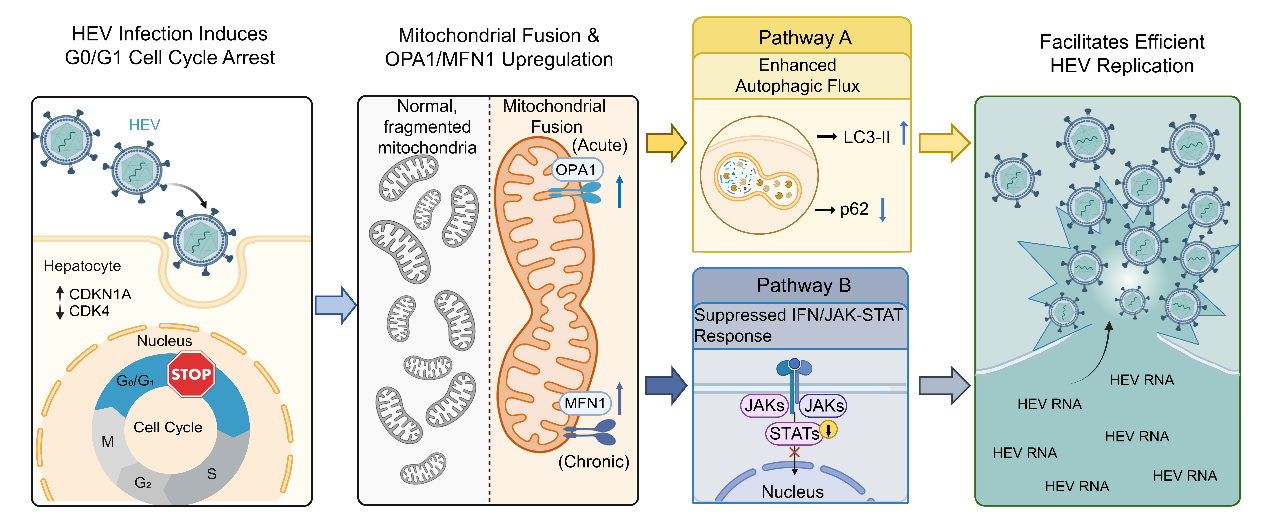
In this study, the researchers found that HEV induces profound mitochondrial structural remodeling characterized by an elongated morphology in both patient liver biopsies and cell culture models. Clinically, elongated mitochondrial profiles in patient livers significantly correlated with disease severity, including elevated aspartate aminotransferase (AST) levels. Mechanistically, this remodeling is driven by a biphasic regulatory program: acute infection preferentially upregulates the inner membrane fusion protein OPA1, whereas chronic infection shifts dependence to the outer membrane mediator MFN1. The researchers also identified that HEV orchestrates a G0/G1 cell cycle arrest to actively enforce and maintain this pro-fusion state. On the one hand, this virus-induced fusion licenses a pro-viral autophagic response required for efficient viral replication. On the other hand, it concurrently dampens interferon-stimulated gene expression to facilitate innate immune evasion.
In conclusion, HEV infection actively co-opts host mitochondrial dynamics to establish a fused mitochondrial network, representing a viral strategy that integrates organelle dynamics, autophagy, cell cycle control, and immune evasion to promote replication. Importantly, genetic ablation of OPA1 or MFN1 reversed mitochondrial elongation and significantly suppressed HEV replication. These findings position mitochondrial fusion machinery as a promising target for therapeutic intervention against hepatitis E.
Read the full research article: Free Radic Biol Med 2026 Mar 10;249(0):260-272. DOI: 10.1016/j.freeradbiomed.2026.03.025