The hepatitis E virus (HEV) is a major cause of acute viral hepatitis worldwide and is primarily transmitted via the fecal-oral route. While HEV replication has been extensively studied in hepatocytes, its interaction with the intestinal epithelium, an early site of viral exposure, remains poorly understood.
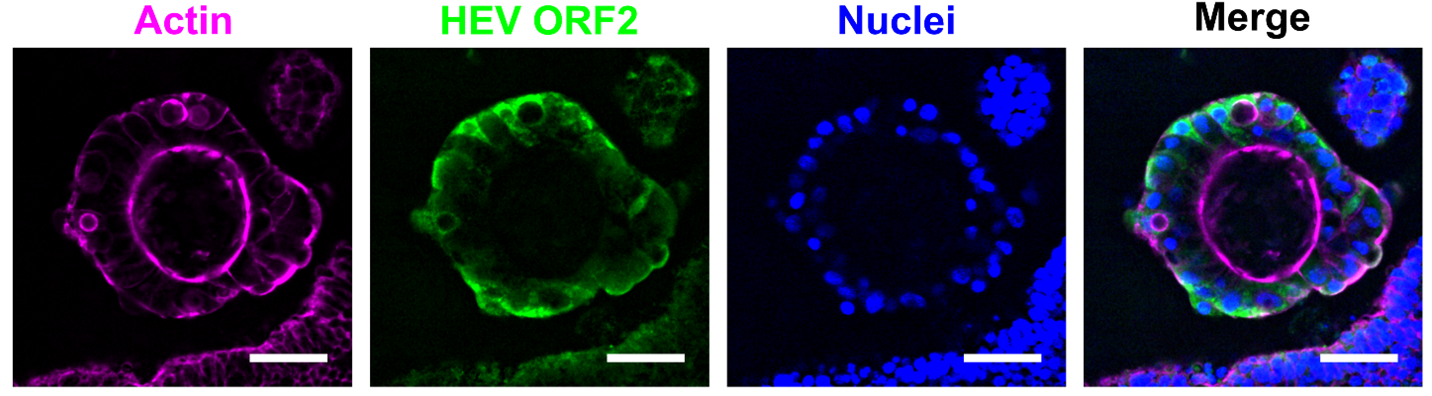
In a recent study by Prallet et al., led by Dr. Viet Loan Dao Thi at Heidelberg University, the authors investigated HEV infection dynamics using human intestinal organoids (hIOs), a physiologically relevant model that recapitulates the cellular diversity and architecture of the intestinal epithelium. Through infection assays, long-term passaging, and single-cell RNA sequencing, they demonstrate that HEV replication was enriched in proliferative crypt-resident cell populations, including intestinal stem cells (ISCs) and transit-amplifying (TA) cells, rather than in fully differentiated enterocytes. These progenitor cells supported sustained viral replication over extended periods, with infection maintained through self-renewal and cell division. Consistent with these findings, HEV RNA was detected in intestinal crypts of an HEV-infected patient.
Single-cell transcriptomic analyses revealed a pronounced cell type-specific antiviral response: enterocytes mounted a robust induction of interferon-stimulated genes (ISGs) following infection, whereas ISCs and TA cells displayed minimal inducible responses despite harboring viral RNA. This differential responsiveness provides an additional explanation for the restricted replication observed in differentiated cells and the enhanced permissiveness of proliferative progenitor populations.
Together, these findings demonstrate that HEV preferentially targets the intestinal crypt compartment and exploits cell renewal to maintain infection. This study establishes human intestinal organoids as a powerful platform to investigate HEV-host interactions and highlights the importance of cellular identity in shaping viral tropism and persistence in the human gut.
Read the full article: Sci Adv. 2026 Apr 3;12(14):eaeb2333. DOI: 10.1126/sciadv.aeb2333