Rat hepatitis E virus (rHEV, Rocahepevirus ratti genotype 1) has recently emerged as a zoonotic pathogen. It has been detected in rat populations in several countries and regions, and accumulating case reports have confirmed its ability to infect humans. However, the prevalence of rHEV exposure in the general population, its clinical impact on different hosts, and the precise routes of transmission remain largely unknown.
A recent study from the University of Hong Kong led by Dr. Siddharth Sridhar addressed this gap by leveraging a large-scale, real-world clinical cohort spanning eight years to systematically characterize the clinical and epidemiological features of human rHEV infection. The findings reveal a disproportionately high risk of clinically relevant rHEV infection for immunocompromised individuals and provide critical insights into this emerging zoonotic pathogen.
Key findings: Immunocompromised individuals require heightened vigilance
The research team systematically screened 2018 patients with hepatitis of unknown cause. Among them, 22 cases (1.1%) were identified with rHEV infection, and 78 cases (3.9%) were diagnosed with swine HEV infection. Compared with HEV infection, rHEV shows a distinctive clinical profile: among the 22 rHEV-infected individuals, 63.6% (14/22) were immunocompromised, whereas immunocompromised individuals accounted for only 28.2% (22/78) of HEV infections during the same period. Most immunocompromised rHEV patients were transplant recipients. Moreover, the study clearly demonstrates that the majority of immunocompromised rHEV patients progress to chronic hepatitis, posing challenges for long-term clinical management. Notably, acute hepatitis associated with rHEV tends to be clinically mild, which may lead to underrecognition or underdiagnosis.
Seroprevalence: Low population exposure, but age-dependent
To assess the level of exposure in the general population, the study tested 8294 serum samples from four groups: healthy individuals, hepatitis C virus carriers, people living with HIV, and solid organ transplant recipients. The overall seroprevalence of rHEV IgG was 0.7% (57/8294), substantially lower than that of HEV IgG at 6.6% (551/ 8294), suggesting limited circulation in the general population. Among the four groups, rHEV IgG seroprevalence ranged from 0.5% to 1.2% (healthy individuals: 0.5%; HIV: 1%; transplant recipients: 1.2%; HCV carriers: 0.9%), but these differences were not statistically significant. Increasing age was the only significant predictor of rHEV seropositivity (odds ratio: 1.03; 95% CI: 1.01–1.05), indicating that exposure risk may accumulate with age.
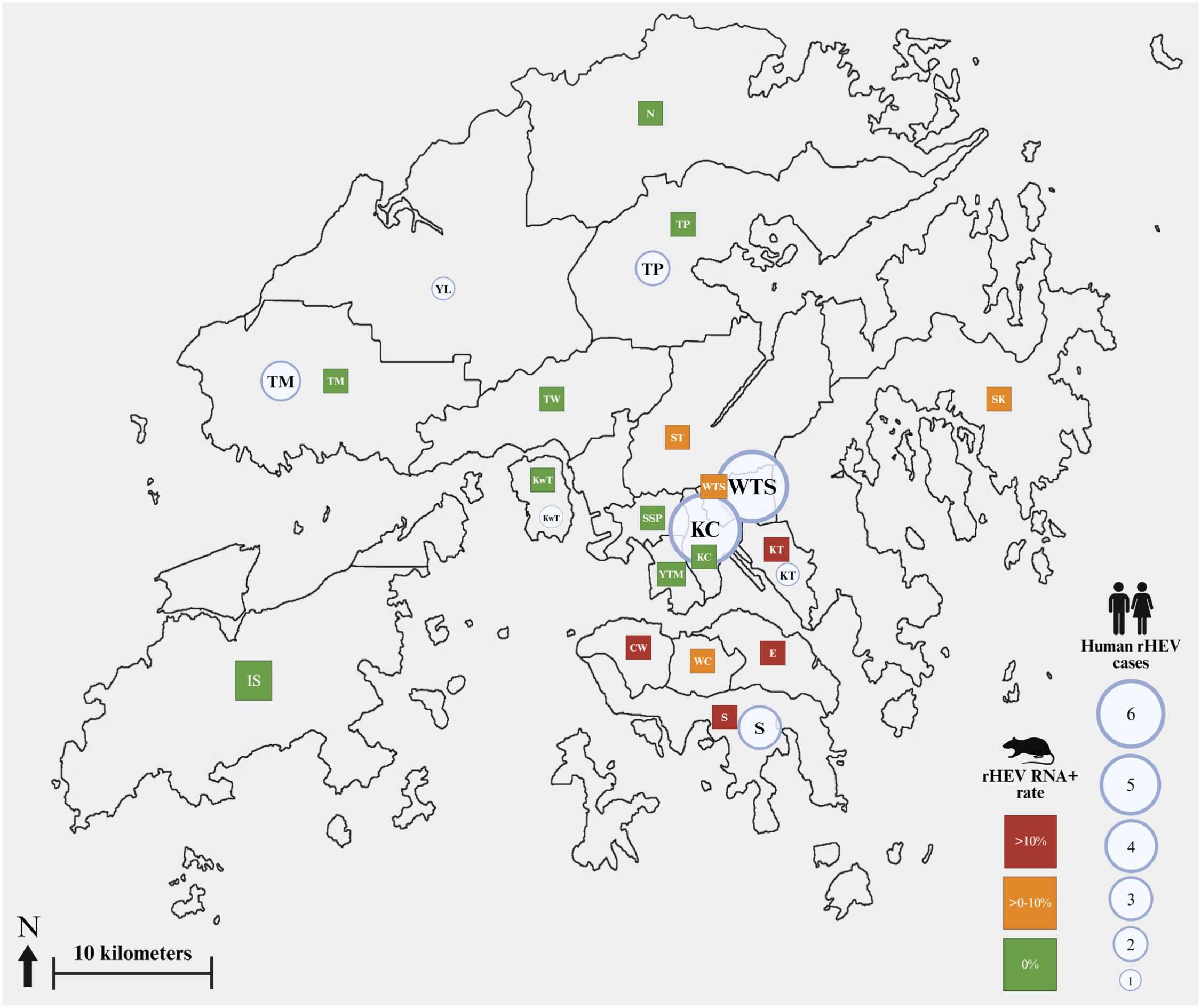
Transmission puzzle: Rats are the source, but how does it spread to humans?
For viral source tracking, the researchers tested commensal rats captured during the same period and detected rHEV RNA. Rat-derived rHEV belonged to two subtypes according to a recently published classification scheme: Ia and IIb1. Only subtype II.b was identical to the strain identified in Hong Kong human cases, providing direct evidence for rats as a natural reservoir. Subtype Ia, which has caused human infections in Europe, was also found in Hong Kong rats but did not spill over to humans locally. However, key questions remain unanswered: What is the exact route of transmission from rats to humans? Is it direct contact, environmental contamination, or food/waterborne transmission? Notably, individuals with bloodborne pathogens did not show higher rHEV seroprevalence, which does not support bloodborne transmission as a major route.
Read the full article published in Journal of Infection: 2026 Jan;92(1):106667. DOI: 10.1016/j.jinf.2025.106667
References
1 .Lo KH, Ho SS, Shun EH, Wu S, Yip CC, Situ J, Ip JD, Chan WM, Yin F, Li Z, To KK, Chan JF, Sridhar S. Enhanced surveillance, subtyping, and host adaptation analysis reveal genotype-wide zoonotic potential of rat hepatitis E virus. J Hepatol. 2026 Jan;84(1):51-61. doi: 10.1016/j.jhep.2025.07.004. Epub 2025 Jul 14. PMID: 40669520.