According to World Health Organization, hepatitis E virus (HEV) caused an estimated 19.47 million cases of acute hepatitis E (AHE) globally in 2021; HEV was responsible for 5.4% of global disability-adjusted life years (DALYs) related to acute hepatitis. Of the eight HEV genotypes (genus Paslahepevirus, HEV-1 to HEV-8), humans are considered as the natural hosts of HEV-1 and HEV-2, whereas the other genotypes have a broad range of animal reservoirs. HEV-1 is the main cause of acute icteric hepatitis in developing countries and causes 20% of mortality in infected pregnant women. While HEV-3 and HEV-4 are zoonotic and account for most sporadic and chronic hepatitis E cases in developed countries.
It is known that HEV-1 is confined to humans, but this notion is challenged by a recent report that Mongolian gerbil, a small rodent can be experimentally infected with HEV-1. An in silico study also suggested that human-associated HEV likely had an origin of rodents. Rodents, the most speciose mammals, often live in close proximity to humans, sharing overlapping habitats, including water sources. This close contact enables wild rodents to transmit major zoonotic pathogens to humans, such as rat HEV (genus Rocahepevirus), which is increasingly detected in humans, causing acute and chronic hepatitis E. Although human-associated HEV is rarely reported in wild rodents. A study from the early 1990s documented a hepatitis E outbreak in humans in Kyrgyz Republic, where HEV (probably HEV-1) was detected in rodent blood sera using immuno-electron microscopy. Despite limited data and no subsequent independent confirmation, this report suggests that wild rodents may play an underrecognized role in the HEV-1 transmission between humans and animals. Given the recent evidence indicating a broader host tropism for HEV-1 than previously assumed, further investigation into additional wild rodent species is warranted.
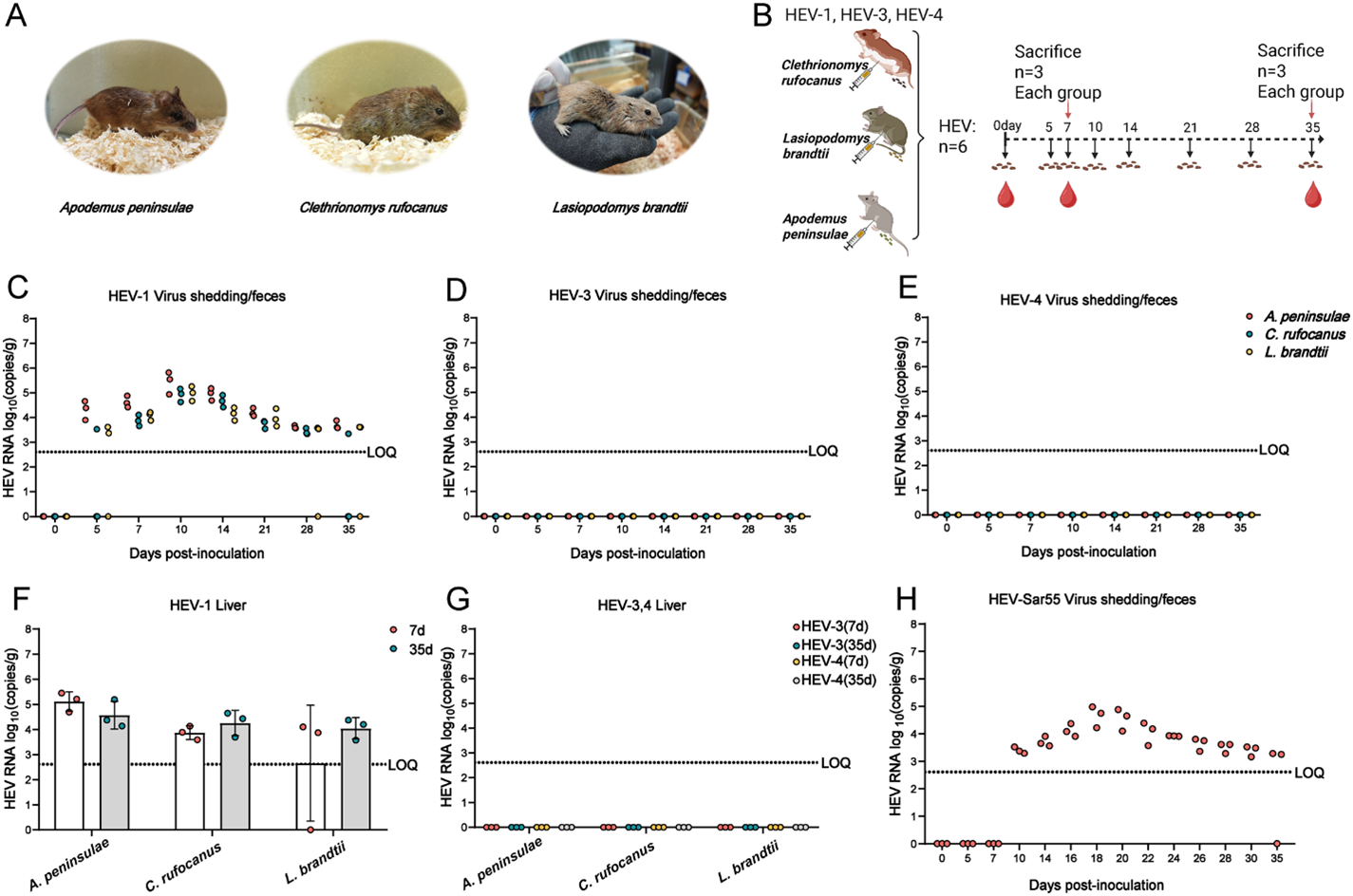
Recently, Dr. Lin Wang, Dr. Xin Yin and Dr. Changyou Xia, from China, domesticated three wild rodent species-Apodemus peninsulae (A. peninsulae), Clethrionomys rufocanus (C. rufocanus), and Lasiopodomys brandtii (L. brandtii), from the wild populations to laboratory breeding. They then investigated their susceptibility to HEV-1 infection. Following HEV-1 inoculation, all three species exhibited detectable fecal HEV RNA shedding and HEV RNA in multiple tissues, including the liver, spleen, kidney, and intestine. Among them, A. peninsulae was more susceptible to HEV-1 when compared to the other two rodent species. To further confirm A. peninsulae’s susceptibility, they used another HEV-1 strain, Sar55 (subtype 1a), and found that A. peninsulae was also susceptible to this strain. In contrast, inoculation with HEV-3 and HEV-4 resulted in no detectable HEV RNA in feces or tissues, indicating genotype-specific susceptibility in these wild rodents.
It is well-recongnized that hepatitis E virus genotype 1 (HEV-1) infects humans only. This study, however, demonstrates that wild rodents—particularly the Apodemus peninsulae—can be infected with HEV-1, exhibiting viral shedding patterns and immune responses similar to those in human infections. The findings suggest that wild rodents may serve as potential natural reservoirs for HEV-1, providing new insights into cross-species viral transmission. The study also confirms HEV-1 can transmit via fecal-oral route and vertical transmission route in Apodemus peninsulae. These discoveries may reshape our understanding of HEV-1, indicating that wild rodents may play a potential role in outbreaks.
Read the full article: PLoS Pathog. 2026 Mar 18;22(3):e1014050. DOI: 10.1371/journal.ppat.1014050